OralTox Certified Training
This certified training program is intended to provide drug test administrators information to better understand the OralTox product and testing procedure. Please read through all of the information within this training program. After completing the training portion, you may test your knowledge with our online certification quiz. Once you have successfully passed the quiz with 100% accuracy, you will receive a personalized certificate of training which you can print for your records.

Product Overview
OralTox is a lateral flow immunoassay, rapid oral fluid test for simultaneous detection of up to 12 drugs. For In vitro diagnostic use for point-of-care cleared for the following drugs: Amphetamine, Cocaine, Marijuana, Methadone, Methamphetamine, Opiates, Oxycodone and Phencyclidine.
Product Features And Benefits
- Patented design with one drug per strip
- Customizable testing panels available
- Observed collections, alleviating adulteration concerns common with urine testing
- Eliminate need for controlled collection sites and same sex collectors
- Built in gravity feed for reliable oral fluid distribution
- Saturation Indicator (Ensures ample oral fluid has been collected)
- Clear and easy to read results
- Easily administered procedure
- Eliminate problems with “shy bladders”
- Device can be sent to the laboratory for confirmation testing without the need to collect an additional sample
- Proven accuracy, sensitivity and reliability
- Fast, accurate results in minutes
- Leading detection of THC parent compound
- Unique QR (Quick response code) ties the device to the donor
- Twist down lid tightens into place to ensure the integrity of the specimen
Storing And Packaging
- Carefully inspect the OralTox pouch to ensure there are no tears, cuts, or damage to the packaging.
- Check the lot number and expiration date. The expiry date is typically 1 year from time of delivery.
- Do not use the device after the expiration date printed on the pouch, example: 2018-04 = April 30, 2018
- OralTox test is to be stored at 36-86°F (2-30°C) in its sealed foil package. If storage temperatures exceed 86° F (30°C), the test performance may degrade.
Components And Test Preparation
- The donor must avoid placing anything in their mouth for at least 10 minutes prior to the collection. This includes, food, drink, gum and tobacco products.
- To aid in collecting enough oral fluid, the donor should be instructed to pool oral fluid prior to collection.
- Open OralTox device pouch in front of the donor
- Request the donor remove the collection swab from the OralTox device.
Specimen Collection
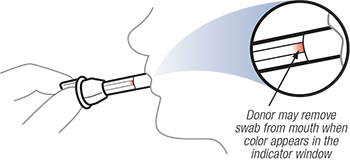
Have the donor sweep inside of mouth (cheek, gums, and tongue) and then hold the swab in mouth until the color fills the indicator window of the collection swab.
Most donors will saturate the collection sponge within 2-4 minutes. Please note that some individuals will experience dry mouth and collection may exceed 4 minutes.
It is very important that the donor does NOT SUCK OR CHEW on the collection sponge or remove the collection sponge from his/her mouth during the collection time, until the indicator changes color.
The collection is completed when color appears on the indication strip inside the collection swab which indicates there is sufficient amount of oral fluid for testing. If after 7 minutes the saturation indicator has not changed color, discard the device and repeat the test.
Test Activation
Note: Administrator supervises the donor throughout the collection process without the need to touch the collection device.
With the test device positioned upright on a FLAT SURFACE, the donor removes the swab from their mouth and inserts the collection swab sponge first into the device, pushing and twisting down until the threaded handle locks into place.
The control line is the uppermost line appearing in each test area. Before reading the test result lines, verify that all control lines have formed. Results from any test without a top control line is invalid and the test must be discarded.

Interpreting Test Results
NEGATIVE results can be read as soon as the test and control lines appear on any test strip (often within 2 minutes).
A negative result is indicated by two colored bands appearing on the membrane. One band appears in the control region (C), and another band appears in the test region (T). The intensity of the test lines may vary. Any line, without regard to intensity, color or size, is a line and indicates a negative result.
PRESUMPTIVE POSITIVE results are to be read at 10 minutes. Interpret test as negative, presumptive positive, or invalid. Do not read test results after 20 minutes. Follow your organizations established protocol for presumptive positive results. After the results have been interpreted, the OralTox device may be thrown in a regular trash receptacle.


Test Your Knowledge By Taking The Certification Quiz
You have now completed the OralTox training. You may go back over any or all of the material above anytime. Once you feel confident with the material presented, you may proceed to the certification quiz by clicking the button below. After you complete the quiz with 100% accuracy, a certificate will be generated on your screen that you may print for your records.
