Premier Bio-Dip Certified Training
This certified training program is intended to provide drug test administrators information to better understand the Premier Bio-Dip product and testing procedure. Please read through all of the information within this training program. After completing the training portion, you may test your knowledge with our online certification quiz. Once you have successfully passed the quiz with 100% accuracy, you will receive a personalized certificate of training which you can print for your records.

Product Overview
The Premier Bio-Dip is a preliminary screening test for the presence of drugs. The Premier Bio-Dip is a qualitative, immunoassay urine based rapid test that can test for multiple drugs simultaneously. The strips are hermetically sealed and flood proof allowing walk-away capabilities to the collector, even when submerged in the sample. The Premier Bio-Dip offers the ability to test for the most relevant drugs of abuse with up to 60-minute result stability.
Product Features And Benefits
- Test for multiple drugs simultaneously
- CLIA-Waived drug test strips available
- Hermetically sealed and flood proof
- 60 minute result stability (Even while submerged in sample)
- Fast results
- Single (specialty) dips available
- Manufactured with automated technical manufacturing and semi-automated assembly
- Proven accuracy, sensitivity, specificity
- Ability to test for relevant drugs of abuse, including: K2/Spice, Tramadol, Fentanyl, EtG, Alcohol, etc.
- Aggressive cut-off levels available
Storing And Packaging
- The Premier Bio-Dip device should be stored at room temperature (15-30 degrees°C/ 59-86 degrees°F).
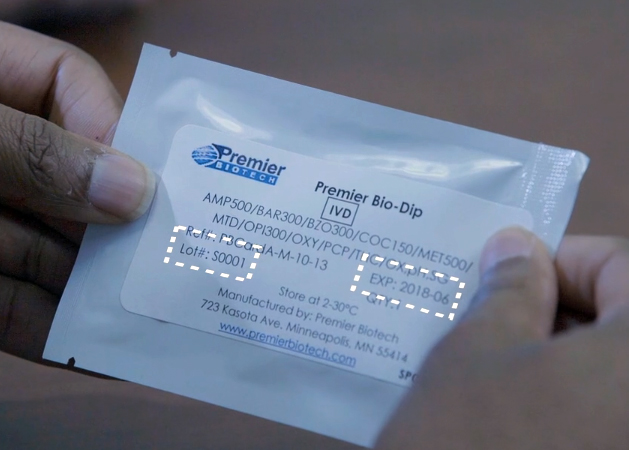
- Carefully inspect the foil pouch to ensure there are no tears, cuts, or damage to the packaging.
- Check the lot number and expiration date. Do not use if past the expiration date. The expiration date of the device is good through the last day of the month noted on the pouch. Example: 2018-06 = June 30, 2018.
Procedure And Specimen Collection
- Have available for use a test results form/record template
- Require donor to present photo ID
- Ask donor to remove any unnecessary outer clothing and empty all pockets
- Secure the collection site, keeping all backpacks and purses out of the restroom
- Turn off water sources (sink faucets)
- Remove all substances that could be used to adulterate specimen (cleansers, disinfectants, soaps, etc.)
- Instruct donor to wash hands in your sight before donating a sample in order to remove any residue that may interfere with the specimen collection.
- Remove the dip card from the sealed pouch.
- Hand the collection cup to the donor and request they provide the urine specimen.
- At this time, check the temperature strip to make sure the specimen is within normal range. The temperature of the specimen should be within 90-100 degrees Fahrenheit. If the temperature of a specimen is not within normal range, the specimen should be considered invalid and another sample should be collected.
- Next, remove the cap from the Premier Bio-Dip to expose the strips. The strips on the Premier Bio-Dip are hermetically sealed inside a protective, flood-proof plastic cover. With the arrows pointing toward the urine specimen, immerse the sample tips vertically in the specimen.
- The Premier Bio-Dip has walkaway capabilities, even when submerged in the sample. This allows the collector to leave the Premier Bio-Dip submerged in the sample or the cap may be replaced.
Results Interpretation
- The Premier Bio-Dip may contain multiple drugs per strip, followed by the individual drug test strips.
- Make sure that the top control line is visible for all test strips, otherwise the test is invalid and the results must not be used.
-
Two lines appearing show a negative result.
-
Negative results may be read as soon as the top control line and bottom test line or lines appear.
-
Any test line regardless of the intensity, color or size, is a line and indicates a negative result. It is normal for line intensities to vary for different drug strips.
- An Inconclusive result (also may be identified as positive, presumptive positive, or non-negative) is indicated by only the top control line present and the absence of a bottom test line.
- Read Inconclusive results at 5 minutes.

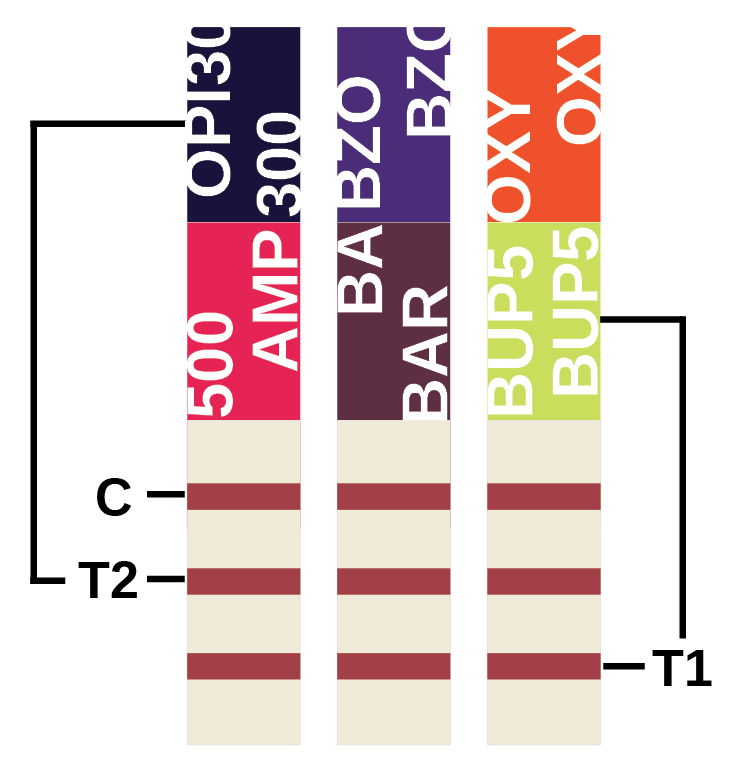
Multi-Strip Interpretation
The multi-drug strips consist of the top control line (C) and two test regions labeled (T1) and (T2) below representing each of the drug test strips. Within the diagram below, (T2) corresponds with OPI, BZO and OXY, while (T1) test lines correspond with AMP, BAR and BUP.

Specimen Validity Testing
The Premier Bio-Dip may come with specimen validity testing. The Premier Bio-Dip specimen validity test is based on the color response of chemical indicators in the presence of adulterants. Specimen validity tests help to determine the integrity of a urine sample. Read the specimen validity test results by visually comparing the color of the reagent pads to the corresponding color blocks on the specimen validity color chart at 3 to 5 minutes. Pad colors that change to a color NOT found listed in the normal range are to be read as abnormal.
Test Your Knowledge By Taking The Certification Quiz
You have now completed the Premier Bio-Dip training. You may go back over any or all of the material above anytime. Once you feel confident with the material presented, you may proceed to the certification quiz by clicking the button below. After you complete the quiz with 100% accuracy, a certificate will be generated on your screen that you may print for your records.
